Opportunity
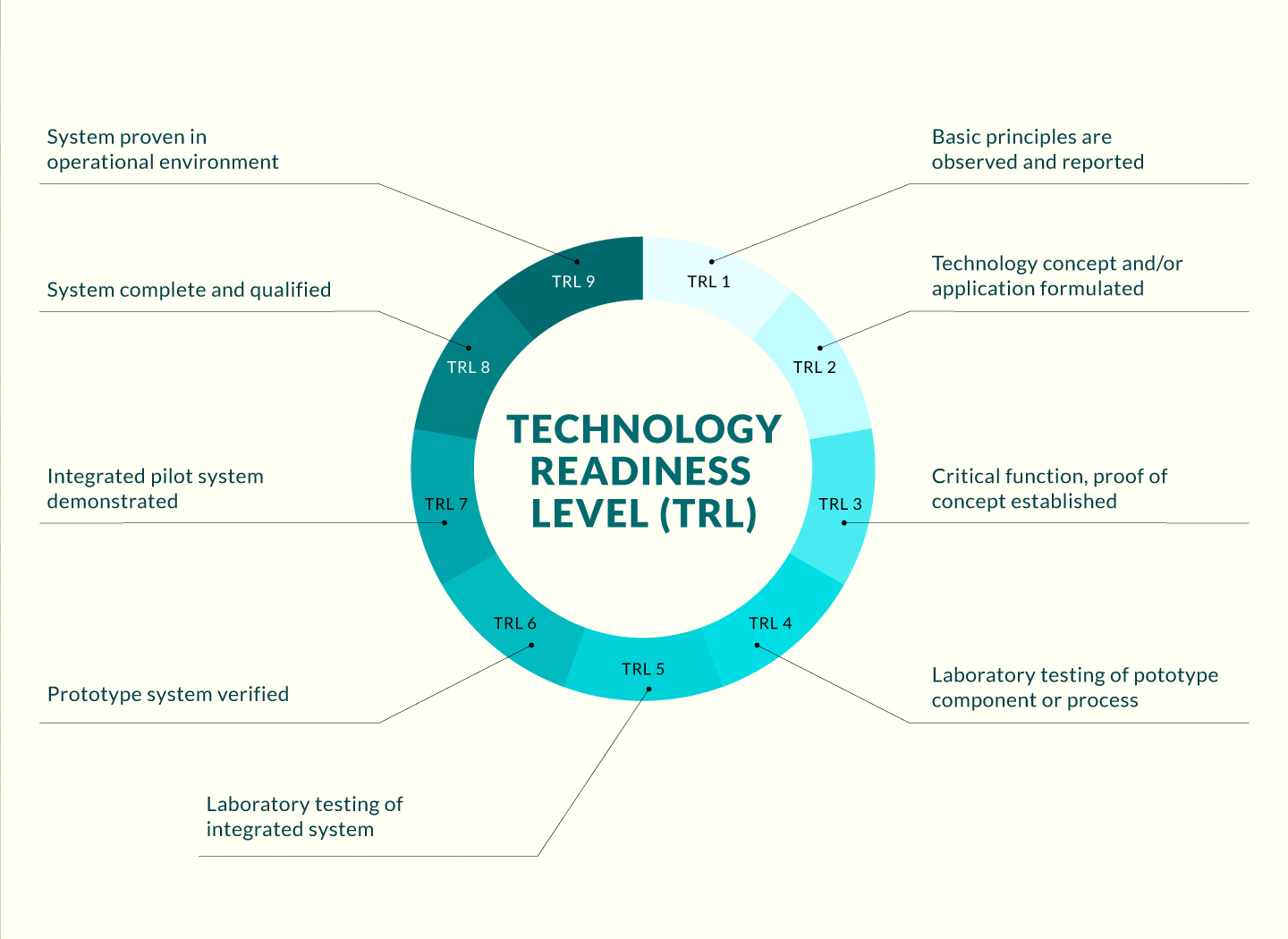
Technologies for mapping dynamic patterns of neural activity over time and space have advanced our understanding of brain function in both health and disease. An important application of these technologies is the discovery of next-generation neurotherapeutics for neurological and psychiatric disorders lacking effective treatments. Moreover, whole-brain imaging of small animals, such as zebrafish, provides an attractive means of bridging the gap between large-scale cellular activity and behavioral responses. Data from this approach can be used to develop drug-screening platforms based purely on complex functional phenotypes.
Technology
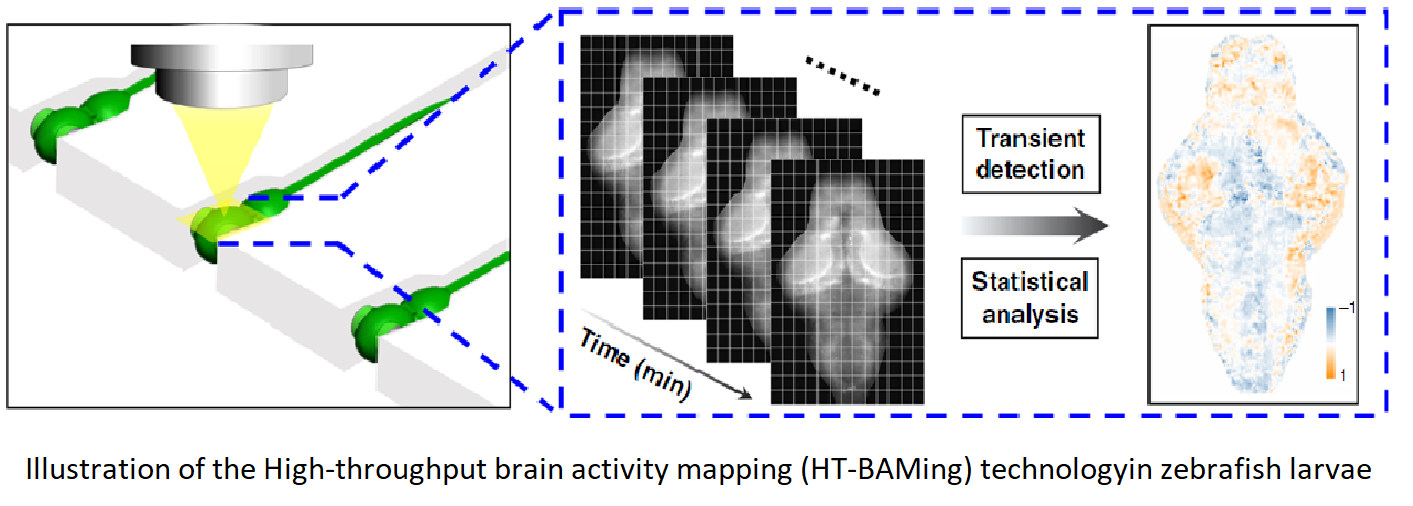
The present technology utilizes functional brain physiology phenotypes and computational bioinfomratics analysis derived from live, nonanesthetized aimnals that have been treated with compounds of interest as an input for predicting the therapeutic potential of neuroactive compounds. Unlike traditional drug screens in relatively simple models, the invented method enables evaluation of a compound's therapeutic potential based on Brain Activity Mapping (BAMs) information derived from drug-treated animals. From a screen of clinically used drugs, we can find intrinsically coherent drug clusters that are significantly associated with known therapeutic categories. Using the BAM-based clusters as a functional classifier, this method allows prediction of specific drugs, such as antiepileptics. Collectively, these BAMs linked to specific compounds provide an experimental framework to advance the field of systems neuropharmacology.
Advantages
- High-throughput of in-vivo drug screening based on brain activity mapping.
- Eicher profiles by machine learning
Applications
- Neuroactive compound drugs screening
- Brain physiology phenotypes